Title
Eli Lilly Atropine Sulfate Tablets No. 205
Author
Eli Lilly & Company, Indianapolis, U.S.A.
Image
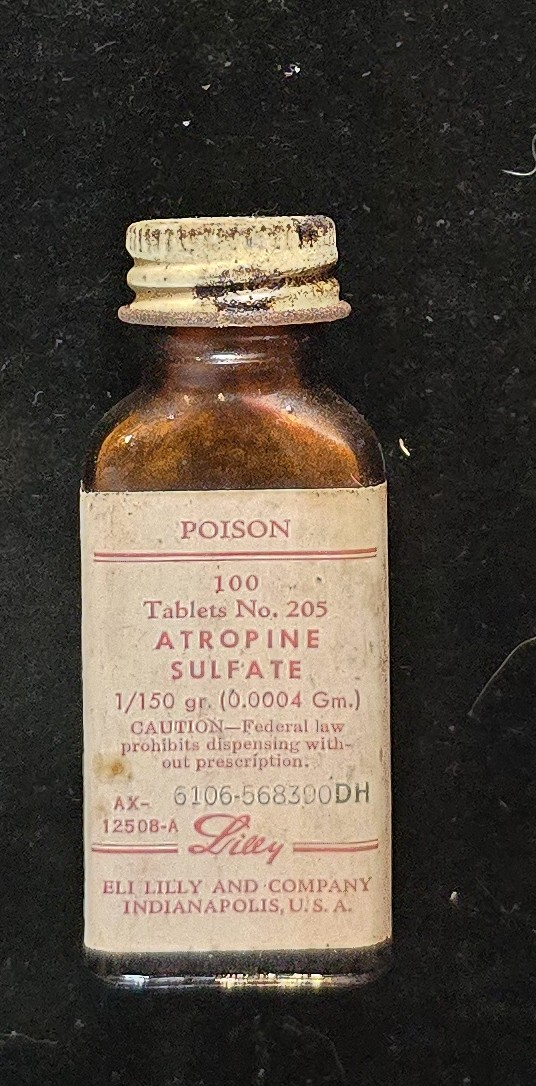
Description
This small amber glass bottle of Atropine Sulfate Tablets No. 205 was manufactured by Eli Lilly & Company of Indianapolis. The label identifies each tablet as containing 1/150 grain (0.0004 gram) of atropine sulfate—a potent anticholinergic alkaloid derived from Atropa belladonna, commonly known as deadly nightshade.
The label bears the red “POISON” warning mandated by mid-20th-century U.S. pharmaceutical regulations, as atropine was considered hazardous even in therapeutic doses. These tablets were typically prescribed to dilate pupils (mydriasis), reduce secretions, counteract organophosphate poisoning, and as a pre-anesthetic agent to inhibit salivation.
This preparation (catalog number AX-12508-A) was part of Lilly’s standardized tablet series produced between the 1930s and 1950s. The amber bottle protected the tablets from light degradation, and the white metal screw cap design is consistent with the company’s packaging style of that period.
Condition
Bottle and label in good condition with minor surface oxidation to cap and slight edge wear. Label is fully legible. Approximately one-third of the original tablets remain inside the bottle, visible through the amber glass.
Gallery
Historical context
Atropine, a tropane alkaloid, has been used since the 19th century as a cardiac stimulant, antispasmodic, and ophthalmic dilator. Its medical history reaches back to belladonna, employed in Renaissance Italy to dilate pupils for cosmetic effect—“bella donna” meaning “beautiful woman.”
During both World Wars, atropine was carried in military medical kits as an antidote for nerve gas and pesticide poisoning, a role it still serves today. By the mid-20th century, manufacturers such as Eli Lilly standardized the compound into precise tablet form, replacing tinctures and crude extracts.
Curious Facts, Ephemera, and Trivia
The “1/150 gr.” dosage predates the modern metric system and illustrates the transitional period between apothecaries’ weight and standardized dosing.
Atropine is on the World Health Organization’s List of Essential Medicines, even today.
The “Lilly” script logo on the label is among the earliest corporate trademarks in U.S. pharmaceutical history.
Overdose symptoms include dilated pupils, flushed skin, confusion, and hallucinations—leading to the old mnemonic: “Red as a beet, dry as a bone, blind as a bat, mad as a hatter.”
Excerpt
“CAUTION—Federal law prohibits dispensing without prescription.”
—Label text, reflecting the 1938 Federal Food, Drug, and Cosmetic Act’s early enforcement of prescription-only medicines.
Why it is in the Cabinet
This bottle represents the intersection of plant-based pharmacology and industrial standardization. It is an artifact of the era when deadly natural alkaloids were tamed into precise, mass-produced tablets—and when “poison” and “medicine” often occupied the same label.
For media or background on the collection, visit the Press & Media Resources page.
Support Dr. Bebout’s Cabinet of Medical Curiosities
If you enjoy the history, the oddities, and the effort, help keep this cabinet open. Every little bit helps preserve and share the strange wonders of medicine's past.
Buy Me a Ko-fi ☕ Buy Me a Coffee ☕ Tip via PayPal 💵